 /PeriodicTableSigFig-NoBG-56a12da75f9b58b7d0bcd00f.png)
Since colligative properties depend only on the number of solute particles, there is no affect of solvent. The activity coefficient is a measure of the degree of ionization of a solute in a solution.Ģ.Does van’t hoff factor depends on solvent?Īns. The Vant Hoff factor is a measure of the degree of dissociation of a solute in a solution. What is the difference between the Vant Hoff factor and the activity coefficient?Īns. It will be one for particles that demonstrate no association or even dissociation. The factor will be greater than one for dissociating solutes. It will always be smaller than one for solutes that show an association. The Van’t Hoff factor gives accurate data on how solutes affect the colligative properties of fluids. Abnormal molar mass occurs when the molar mass is less than or more than the predicted value. of moles available in a solution following solute association or even dissociation. Readings of molar mass in this scenario will be higher than expected. Since the number of solute particles is reduced as a result of their interactions, colligative characteristics are also reduced.Due to the inverse relationship between molar mass and colligative properties, we anticipate a smaller final outcome.The dissolution of solute components into many ions increases the total particle content and hence enhances the colligative features of the mixture.The following table summarizes the consequences of a solute’s dissociation or even association on the Van’t Hoff factor.Īn irregularity in molecular mass occurs in the following cases:.Sodium chloride breaks down into Cl- and Na+ ions when exposed to water. Dissociation is the process by which a substance is split up into its constituent ions.Carboxylic acids show a high degree of association through hydrogen bonding. The combining of 2 or more substances to produce a single object is known as an association.A greater divergence is seen at higher ion charges. In the case of electrolytic fluids, it is important to note that the measured factor will be smaller than predicted. Jacobus Henricus Van’t Hoff, the first recipient of the Nobel Prize in Chemistry, is honoured with the naming of this constant. 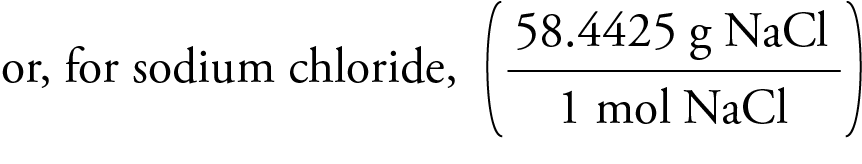
When dissolving in water, the number of ions ( i ) remains constant regardless of how many atoms (N) of the substance are dissolved. 
When something that isn’t electrolytic dissolves in water, the value of i will typically always equal 1. A material’s degree of association or dissociation in a fluid is the relevant factor. To make sense of this variation in setup, the Vant Hoff factor is employed. This occurs because the arrangement of atoms in a substance is not constant. Because the actual molar mass of a substance is not always equal to the sum of the atomic masses of the atoms making up the substance, a quantity called the Vant Hoff factor is employed to account for this discrepancy.Van’t Hoff factor is used in such cases to find the actual molar mass. Among the colloquial features are a higher osmotic pressure, a lower vapour pressure, a lower freezing point, and a higher boiling point. They are calculated using the colligative characteristics. Let knowledge be the cure.Molar masses that are calculated to be too high or too low than the actual molar mass are deemed abnormal molar mass. Powered by django-wiki, an open source application under the GPLv3 license. This article was last modified: July 27, 2022, 8:44 a.m. You can remember that the little m should be at the top, because it looks like a bird which fly high above everything else. When you cover the item you want to calculate, you have the equation written right there. One way to do that is to write it up in a triangle like this: These three equations are very useful for many purposes in chemistry and it can be practical to find a mnemonic to memorize it. But when you look at the unit (g/mol) you can see that molar mass can also be calculated if you know the mass of a substance (g) and how many moles that corresponds to (mol): M(g/mol) = m(g)/ n(mol)īy rearranging this equation you can also calculate how many grams, a certain number of moles should weigh, by isolating m in the equation: m(g) = n(mol) * M(g/mol) One mole of a substance will have the same value as the molecular mass of that substance, which can be calculated from the atomic weights of its constituent atoms. The molar mass, M, is the mass in grams per one mole (g/mol or gᐧmol -1) of a substance. Where m is the mass and M is the molar mass of the given substance. The number of moles, n, of a substance can be found by using the following equation n(mol) = m(g) / M(g/mol) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
